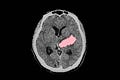
Minimally-invasive surgery to evacuate intracranial hemorrhages improved outcomes (ENRICH trial)
BrainPath device saved functional independence, among a highly selected group of patients
Patients with certain spontaneous intracerebral hemorrhages treated with minimally invasive surgery had improved functional outcomes compared to those treated with usual care, the ENRICH randomized trial showed.
Treatment options for spontaneous intracerebral hemorrhage have remained severely limited, with little improvement in outcomes over decades. Craniotomy is generally considered a salvage therapy reserved for patients who appear near death; it has never been shown to improve functional outcomes.
Numerous minimally invasive surgery techniques for intracerebral hemorrhage have been produced and tested over the past decade. In the ENRICH trial, investigators used a new surgical tool called the BrainPath device (Nico Corporation).
The BrainPath device uses imaging guidance to allow a neurosurgeon to navigate between the folds of the brain, producing minimal damage to healthy brain tissue while advancing a catheter that can access the area of supratentorial hemorrhage. A separate device called Myriad performs the actual suction of hemorrhage and hematoma.
The ENRICH trial used an adaptive Bayesian enrollment design, as is becoming more common. Three hundred patients were enrolled at 37 U.S. centers, out of more than 11,000 screened over 5 years. All had supertentorial spontaneous intracranial hemorrhages, in the basal ganglia or lobar (superficial) locations, estimated between 30 ml and 80 ml in volume. Enrollment was switched to include only patients with lobar hemorrhages after 175 patients, when it appeared that basal ganglia bleeds were not being helped (a prespecified futility trigger).
Subjects were randomized to either minimally invasive surgery with the BrainPath/Myriad system within 24 hours, or usual care. Surgery was highly effective at removing hematoma (with 15 ml or less remaining after evacuation in most patients). The trial was funded by the Nico corporation; authors stated Nico had no involvement in trial design, data analysis or manuscript preparation.
Patients treated with minimally invasive surgery experienced significant benefits:
After 30 days, only 9% of patients treated with minimally invasive surgery had died, compared with 18% in the usual care group. (Those benefits had largely disappeared at 6 months, when 20% of patients treated minimally invasively had died, compared to 23% with usual care.)
Surgically-treated patients who survived had durable improvements in functional outcomes compared to usual-care patients, with utility weighted modified Rankin scale scores of 0.458, compared to 0.374 in the control group.
Far fewer patients treated with minimally invasive surgery eventually required decompressive hemicraniectomy (3.3% vs 20%).
About 3% of surgically treated patients had postoperative rebleeding resulting in worsened neurologic outcomes.
The long-term functional outcome improvement was certainly not huge, but it was clinically significant. The average patient in this trial hovered in a status between modified Rankin scores 3 and 4 (0.65 and 0.33 on the utility-weighted scale). That’s the difference between being able to walk and attend to personal needs independently, or not.
In other words, this new technique wasn’t primarily “successful” at allowing more people to survive their brain hemorrhages into permanent disability. It genuinely helped some people preserve their independence, and others to lose less of theirs.
For example, 47 patients treated with the new device survived for six months with only slight disability, vs. 26 treated with usual care. This isn’t statistically robust enough to draw firm conclusions, but out of 300 patients with such poor expected outcomes, it’s enough to take optimistic note.
The benefits of surgery were even larger in lobar hemorrhage patients—with an overall benefit persisting even when including the 175 patients with basal ganglia hemorrhage, who were not helped.
This was an open label (unblinded) trial, so the usual caveats apply about the possibility of unmeasured treatment differences between groups. The patients were highly selected, as evidenced by the slow enrollment and small sample size.
Conclusion
Minimally invasive surgery meaningfully improved functional outcomes for lobar (superficial) intracranial hemorrhages—the first unqualified good news in decades for this often devastating condition. At least five additional randomized trials testing minimally invasive surgery for intracranial hemorrhage are currently in process.